 Four electrons are used so far, and there are 16 – 4 = 12 electrons remained.Ĥ. Total number of valence electrons: 4 (C atom) + 2×6 (2 O atoms) = 16Īlways DOUBLE CHECK: In the correct Lewis structure, the total number of electrons involved (bonding plus non-bonding electrons) must be equal to this number, less or more are both incorrect!!Ĭarbon atoms are always central, so the skeletal structure is: O - C - Oģ. 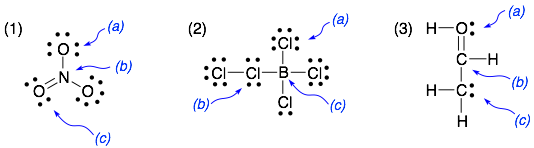
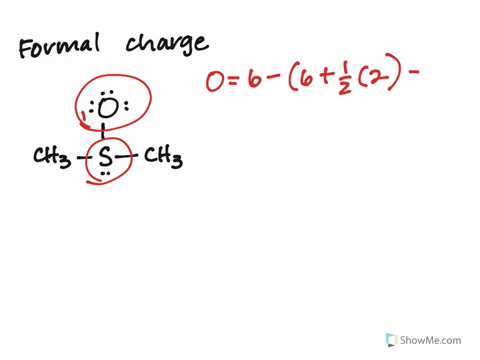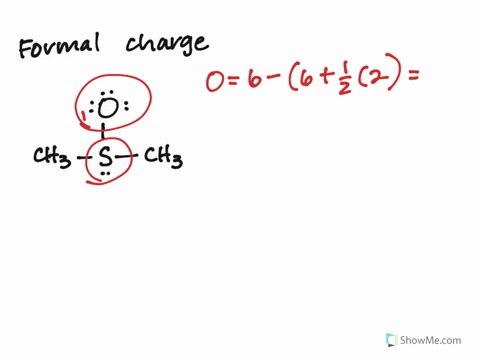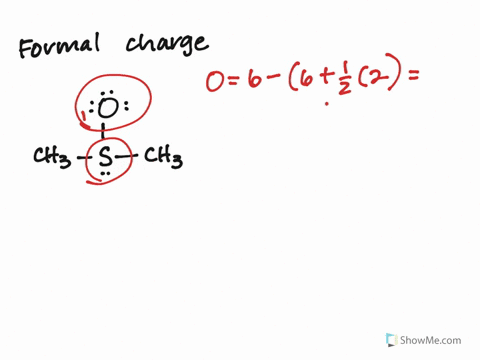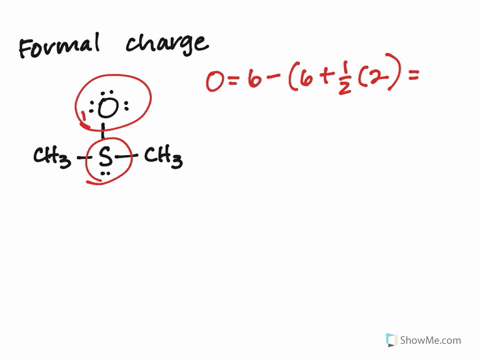
Calculate the Formal Charges on all atoms and label the non-zero formal charges in the structure:įormal Charge on an atom = No. If not, then complete the octets of all central atoms by moving lone pairs from terminal atoms to form multiple bonds.ħ. If you have used up all the valence electrons to complete octets for all the atoms, you are done.Ħ. Using the remaining valence electrons, complete the octets of the terminal atoms first, then complete as many as possible for the central atoms.ĥ. 
For each single bond, subtract two electrons from the total number of valence electrons.Ĥ.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
